|
|
 发表于 19-11-2010 06:29 AM
|
显示全部楼层
发表于 19-11-2010 06:29 AM
|
显示全部楼层
after combustion...the gases contain unreacted oxygen and carbon dioxide produced.
volume of carbon dioxide produced:134-52=82 cm^3
1 mol of methane produced 1 mol of carbon dioxide.(refer to your own balanced equation.)
let the initial volume of methane be x,
1 mol methane produces 1 mol of co2,
then x cm^3 of methane also produced x cm^3 of co2.
1 mol of ethane produced 2 mol of carbon dioxide.(refer to your own balanced equation)
the volume of initial volume of mixture is 50 cm^3,so the volume of ethane gas is (50-x) cm^3
1 mol of ethane produced 2 mol of co2.
(50-x) cm^3 of ethane produced (82-x)cm^3 of co2...
*total co2 produced is 82cm^3,methane produced x cm^3...then the rest of the co2 produced by ethane.
cross and multiply then get x=18 cm^3,the volume of methane. |
|
|
|
|
|
|
|
|
|
|
|
 发表于 21-11-2010 08:26 PM
|
显示全部楼层
发表于 21-11-2010 08:26 PM
|
显示全部楼层
Will copper react with dilute sulphuric acid?
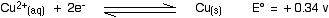
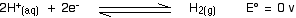
If I apply Ecell = Ecathode - E anode, I will get a positive value, but why say the reaction not occuring spontaneously?? |
|
|
|
|
|
|
|
|
|
|
|
 发表于 21-11-2010 10:04 PM
|
显示全部楼层
发表于 21-11-2010 10:04 PM
|
显示全部楼层
the E value should be negative...
take notes:copper not copper ions...looks from right is -0.34 v |
|
|
|
|
|
|
|
|
|
|
|
 发表于 21-11-2010 11:05 PM
|
显示全部楼层
发表于 21-11-2010 11:05 PM
|
显示全部楼层
|
what do you mean?? Ecathode - E anode straight apply the details given ma.In this case cathode is Cu ma |
|
|
|
|
|
|
|
|
|
|
|
 发表于 22-11-2010 06:48 AM
|
显示全部楼层
发表于 22-11-2010 06:48 AM
|
显示全部楼层
本帖最后由 nkrealman 于 22-11-2010 08:31 AM 编辑
i think u confuse with the words cathode or anode.only copper and acid is provided.
we drop the copper into the acid and predict either reaction will occur or not.
copper never be cathode.cathode mean the reduction take places.thus,copper will receive electrons and become negative ions..it couldn't happen in normal situation..and the E values is not given |
|
|
|
|
|
|
|
|
|
|
|
 发表于 25-11-2010 09:20 PM
|
显示全部楼层
发表于 25-11-2010 09:20 PM
|
显示全部楼层
请问要怎样看得出化学作用后的产物是液体、固体,还是?
燃后,FeSO4 和 Fe2(SO4)3有什么差别?
我的che不好,很少及格……嘿嘿~现在算是从头学起了! |
|
|
|
|
|
|
|
|
|
|
|
 发表于 25-11-2010 09:34 PM
|
显示全部楼层
发表于 25-11-2010 09:34 PM
|
显示全部楼层
本帖最后由 nkrealman 于 25-11-2010 09:36 PM 编辑
回复 729# 邪魅
很好的问题,我好像从来没想过这个问题 organic products 跟 h20 是 liquid organic products 跟 h20 是 liquid
inorganic ,也就是salt 你要看它是不是 soluble,是的话变成 aqueous,不是就是solid...form 4 chemistry salt那个chapter有提到一些。
至于gas就很容易看吧...
FeSO4 和 Fe2(SO4)3都是ionic products
FeSO4 是 Fe2+
Fe2(SO4)3 是 Fe3+ |
|
|
|
|
|
|
|
|
|
|
|
 发表于 29-11-2010 10:12 AM
|
显示全部楼层
发表于 29-11-2010 10:12 AM
|
显示全部楼层
回复 729# 邪魅
这个要看它的特性,比如说Fe3+通常都是brown colour的,而Fe2+是green colour的。。(如果我没记错的话,如果我错了请各位大大纠正我的过错,谢谢。)然后假设react后solution的colour有变动,那就可以看得出来是从什么变去什么。。 |
|
|
|
|
|
|
|
|
|
|
|
 发表于 3-12-2010 12:30 PM
|
显示全部楼层
发表于 3-12-2010 12:30 PM
|
显示全部楼层
勤力的学生应该都不上网了,不过我还是很担心的问一句,pp2第九题的mechanism是sn2还是sn1?
我放sn2可是题目给的graph应该不是sn2的 |
|
|
|
|
|
|
|
|
|
|
|
 发表于 3-12-2010 12:33 PM
|
显示全部楼层
发表于 3-12-2010 12:33 PM
|
显示全部楼层
勤力的学生应该都不上网了,不过我还是很担心的问一句,pp2第九题的mechanism是sn2还是sn1?
我放sn2可是题 ...
henggi 发表于 3-12-2010 12:30 PM 
有些勤力的是网上学习的。。。 |
|
|
|
|
|
|
|
|
|
|
|
 发表于 3-12-2010 12:38 PM
|
显示全部楼层
发表于 3-12-2010 12:38 PM
|
显示全部楼层
google search了一下,sn1 来的。
哈哈哈哈哈哈哈哈哈 ,最少十分飞了 ,最少十分飞了 |
|
|
|
|
|
|
|
|
|
|
|
 发表于 11-12-2010 11:09 AM
|
显示全部楼层
发表于 11-12-2010 11:09 AM
|
显示全部楼层
我后悔没读inorganic的 |
|
|
|
|
|
|
|
|
|
|
|
 发表于 29-12-2010 09:16 PM
|
显示全部楼层
发表于 29-12-2010 09:16 PM
|
显示全部楼层
|
请问下,nucleon number是不是就等于relative atomic mass? |
|
|
|
|
|
|
|
|
|
|
|
 发表于 30-12-2010 03:26 PM
|
显示全部楼层
发表于 30-12-2010 03:26 PM
|
显示全部楼层
|
|
|
|
|
|
|
|
|
|
|
 发表于 30-12-2010 08:38 PM
|
显示全部楼层
发表于 30-12-2010 08:38 PM
|
显示全部楼层
|
|
|
|
|
|
|
|
|
|
|
 发表于 14-1-2011 09:56 PM
|
显示全部楼层
发表于 14-1-2011 09:56 PM
|
显示全部楼层
谢谢。
我老师给我的练习题,我完全看不明白,请告诉我该怎样做,他要求什么! 谢谢!
Suggest a possible identity for each organic compound below. explain reason n account(wat's mean is it?) for the observation.give balanced equation for reaction that occur where approopriate
a)P,molecular formula C7H8 don't readily decolourise aq Br, but on boiling under reflux with aq KMnO4 the latter is decolourised and compound
Q,molecular fomula C7H6O2 is formed.
b)R,C7H7Cl, on warming with aq NaOH,cooling the resulting mixture, acidifying with nitric acid, then add aq silver nitrate gives a white percipitate. |
|
|
|
|
|
|
|
|
|
|
|
 发表于 14-1-2011 10:25 PM
|
显示全部楼层
发表于 14-1-2011 10:25 PM
|
显示全部楼层
KMnO4 是 oxidising agent.
1.oxidising agent 会对Organic compound怎样呢?
2.KMnO4 只会 decolourise 有 double bond的 organic compound? |
|
|
|
|
|
|
|
|
|
|
|
 发表于 15-1-2011 01:21 AM
|
显示全部楼层
发表于 15-1-2011 01:21 AM
|
显示全部楼层
|
2-bromo-3-methylbutane is primary or secondary haloalkane? |
|
|
|
|
|
|
|
|
|
|
|
 发表于 15-1-2011 07:38 AM
|
显示全部楼层
发表于 15-1-2011 07:38 AM
|
显示全部楼层
回复 740# 邪魅
Oxidised那个organic compund咯
也不一定,在Physical chemistry也会出现KMnO4这个oxidising agent |
|
|
|
|
|
|
|
|
|
|
|
 发表于 15-1-2011 07:39 AM
|
显示全部楼层
发表于 15-1-2011 07:39 AM
|
显示全部楼层
回复 741# 邪魅
secondary haloalkane
华语 |
|
|
|
|
|
|
|
|
|
| |
 本周最热论坛帖子 本周最热论坛帖子
|